pE3.1
Shuttle plasmid for the construction of bipartite adenovirus vectors using the AdenoQuick1.0 system
- Replication-deficient E1- and E3- substituted adenovirus vectors (bipartite, dicistronic adenovirus vectors)
- Insertion of heterologous sequences in place of the E3 region of the Ad5 genome
- "Armed" conditionally replicative adenovirus vectors (CRAds)
- E4 region WT or deleted
- WT Ad5, Ad5/35, or Ad5/3 fiber
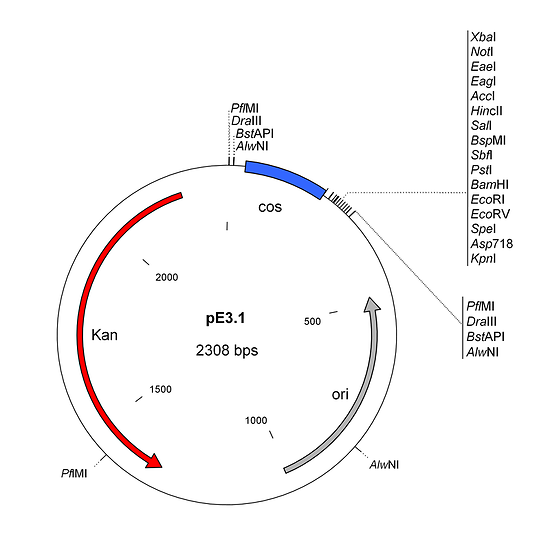
pE3.1 is a shuttle plasmid designed for inserting expression cassettes in place of the E3 region of the Ad5 genome, in combination with the pE1.2 and AdenoQuick1.0 plasmids (pAd292, pAd328, ...). It contains a multiple cloning site (MCS) adjacent to a 178 bp-long λ cos site. Expression cassettes inserted into the MCS should contain a promoter, coding sequence and a polyA signal. The sequences encompassing the cos site and the MCS are flanked by two sets of restriction sites AlwNI, BstAPI, DraIII and PflMI which generate non-symmetrical sticky ends useful for directional cloning. pE3.1 can also be used in combination with plasmids pAd328 and pE1.2 to construct "armed" conditionally replicative adenoviruses (CRAds) containing a heterologous promoter in front of the E1a TATA box, and a transgene in place of the E3 region. It can be used also in combination with plasmids pAd363 and pE1.2 to construct ΔE1/E3/E4 vectors with maximum 8.7 kb cargo capacity.
| Product | Cat# | Size | Price | |
|---|---|---|---|---|
pE1.2Shuttle plasmid for the construction of bipartite adenovirus vectors using the AdenoQuick1.0 system |
Cat# :
QP-02 |
20 ug |
$220.00 |
|
pAd292Ad5 backbone for the construction of replication-deficient E1/E3-deleted dicistronic (a.k.a. bipartite) adenovirus vectors using the AdenoQuick1.0 system |
Cat# :
QR-03 |
10 reactions |
$450.00 |
|
pAd363Ad5 backbone for the construction of replication-deficient E1/E3/E4-deleted bipartite adenovirus vectors using the AdenoQuick1.0 system |
Cat# :
QR-06 |
10 reactions |
$475.00 |
|
pE3-CMVShuttle plasmid for inserting CMV-driven expression cassettes in place of the E3 region of Ad5 adenovirus vectors using the AdenoQuick1.0 system |
Cat# :
QP-17 |
20 µg |
$220.00 |
-
What is a cosmid?
A cosmid is a large plasmid that was generated by infecting E. coli with bacteriophage lambda. Cosmids are really great tools to construct recombinant adenoviruses because their minimum sizes (~39 kb) accommodates the 36 kb adenovirus genomes almost perfectly. When you construct a recombinant adenovirus genome in E. coli using cosmid technology, you are almost sure that every colony carries a full-size genome, and not a smaller plasmid that lost chuncks of the adenovirus genome by DNA recombination.
-
How difficult is it to construct a cosmid?
Constructing a cosmid is not more difficult, more time-consuming nor more expensive than constructing a plasmid.
Here are the steps:
- Ligate your DNA fragments (2-3 hours at R.T. or overnight at 16 ºC)
- Package of the ligation products into lambda phage heads: simply add 2 uL ligation reaction to 6 uL packaging extract (provided in the kit) and incubate at 30 ºC for 90 min.
- Infect E. coli with the packaged lambda heads: 30 min at 37 ºC.
- Streak on petri dish supplemented with LB + antibiotics - incubate overnight at 37 ºC.
- Grow colonies in 2 mL LB/antibiotics (< 12 hrs at 37 ºC)
- Purify the cosmid DNA (alkaline lysis method) and analyze by restriction digestion.
-
Which enzyme should I use to linearize my cosmid before transfecting it into helper cells? PacI or SwaI?
Both PacI and SwaI are rare-cutting restriction enzymes that recognize 8 bp-sequences. Both of them are flanking both ends of the recombinant adenovirus genome contained in your cosmid. You should make sure that the enzyme that you will use to linearize your cosmid does not cut inside your transgene. If neither PacI nor SwaI cuts inside your transgene, there is really no preference. Because the PacI site is immediately flanking the start of the adenovirus genome, the DNA ends generated by PacI resemble the most the ends obtained from deproteinised virion DNA and might therefore be more efficient in promoting virus replication. In practice however, no difference in the time needed to recover the virus after DNA transfection into 293 cells is observed between both settings. Virus plaques can appear as early as 4 days after transfecting PacI- or SwaI-digested DNA into 293 cells.
-
What will happen if I do not digest the cosmid DNA with PacI or SwaI prior to the transfection?
You will most likely not get any virus plaque. Linearizing the cosmid is important to release the ITRs that serves as origin of replication for the adenovirus DNA polymerase.
-
How do I choose the status of the E3 region: wild-type or deleted?
In the adenovirus replication cycle, the expression of the E3 region helps the virus evading the host immune system. This region is not essential for virus replication in vitro and therefore can be deleted in order to construct adenoviruses containing longer transgenes, up to 7.7 kb.
Therefore, if you are using the most common 3.2 kb E1 deletion, and:
- if your expression cassette (= promoter + coding sequence + polyA signal) is smaller than 5.0 kb, you can use adenovirus vectors with either wild-type or deleted E3 region.
- if your expression cassette is larger than 5.0 kb but smaller than 7.7 kb, you must use E1/E3 deleted vectors.
- if your expression cassette is larger than 7.7 kb, you must consider E1/E3/E4-deleted adenovirus vectors.
Notes:
- In some applications such as oncolytic adenovirus vectors (CRAds), it might be desirable to retain the entire E3 region, or increase the expression of some E3 products: for instance, the adenovirus “death protein” E3-11.6K, which facilitates the release of viral particles from infected cells, or gp19K, whose constitutive expression reduces the host cytotoxic T cell response against the vector and increases the persistence of transgene expression on its own but possibly not in the context of constitutive expression of the entire E3 region.
- The E3 region can also be used as a location to insert a second transgene, independent from the one inserted in the E1 region.
-
How do I calculate the maximum cargo capacity of an adenovirus vector?
You simply sum up the sizes of the deletions present in your vector and add 1.8 kb. For instance, let us suppose that you are considering using an Ad5 backbone characterized by a 3.2 kb E1 deletion (psn 354-3510) and a 2.7 kb BglII E3 deletion (psn 28133-30818). Your vector will be able to package 3157 bp (= 3510 - 353) + 2686 bp (= 30818 - 28132) + 1800 bp = 7.6 kb.
The additional 1.8 kb is the extra DNA that Ad5 capsids can package in addition to their 35,935 bp-long genome (Bett et al, J. Virol. 1993; 67: 5911-21).
-
What is the maximum cargo capacity for the AdenoQuick and AdenoZAP system?
For the AdenoQuick1.0 system, the current maximum cargo capacity is achieved with E1/E3/E4-deleted pAd362. The vector allows for inserting 8.9 kb foreign DNA into the E1 region.
For the AdenoQuick2.0 system, adenovirus vectors can be constructed, in which up to 11.2 kb foreign DNA can be inserted. It is done by combining the largest E1 deletion (3157 bp in shuttle plasmid pAd1127-02) with the largest E3 deletion and the hybrid Ad5/35 fiber ( 2686 bp + 756 bp in pAd1129-06), and the largest E4 deletion (2815 bp in shuttle plasmid pAd1130-03), plus the extra 1.8 kb that adenovirus capsids can accomodate in addition to the WT 36 kb genome (Bett et al, 1993. J. Virol. 67: 5911-21).
For the AdenoZAP system, the current maximum cargo capacity of 9.6 kb is obtained with AdenoZAP1.4.
-
I am using the AdenoQuick1.0 system. I am ready to digest the shuttle plasmid with AlwNI, BstAPI, DraIII or PflMI. What is the difference between these enzymes?
AlwNI, BstAP1, DraIII, PflMI and SfiI are enzymes that recognize interrupted palindromes. The sequences that separate the two parts of the palindrome can be chosen arbitrarily. Upon cutting, they will all create 3 nt-long sticky ends with a 3' extension. In the AdenoQuick1.0 plasmids and their shuttle plasmids pE1.1, pE1.2 and pE3.1, we have designed these sites in a specific way: there are 4 sets of AlwNI, BstAPI, DraIII and PflMI sites (they are color-coded in Figure 1 and Figure 2). Within one set, all four enzymes generate the same sticky end. For instance the AlwNI, BstAPI, DraIII and PflMI sites in pE1.2, located downstream from the Kan-resistance gene (coded in yellow in Figure 2), all generate an AGA 3' extension as sticky end. This sticky end cannot be ligated with the sticky ends generated by the same enzymes at other places (such as the blue, green and pink sets shown in Figure 2). However this AGA sticky end can ligate with the sticky end generated by one of the SfiI sites present in the pAd292 (AdenoQuick13.1) vector (also coded in yellow). It does not matter which enzyme you use to cut pE1.1, pE1.2 or pE3.1, as long as it does not cut inside your expression cassette. You can use the same enzyme or not for pE1.2 and pE3.1, the result will be almost exactly the same (a few nucleotides difference).
-
Which facilities are required to work with adenovirus vectors?
The National Institute of Health has designated adenovirus as Level 2 biological agent. For most applications, working with adenovirus requires therefore a Biosafety Level 2 (BL2) facility. The NIH guidelines for research involving recombinant DNA molecules stipulate also that experiments which are likely to either enhance the pathogenicity (e.g. insertion of a host oncogene) or to extend the host range (e.g. introduction of novel control elements) of viral vectors under conditions that permit a productive infection should be performed in BL3 facilities.
A BL2 laboratory should contain:
- A warning sign on the entrance door limiting the access to authorized persons only. The sign should identify the agent, list the name and phone number of the lab director or other responsible person, and indicate any special requirement for entering the lab.
- A Class II biological safety cabinet. A Class II cabinet is a ventilated cabinet for personnel and product protection having an open front with inward airflow for personnel protection, and a HEPA filtered mass recirculated air flow for product protection. The face velocity of the inward flow of air through the full-width open front is 75 feet per minute or greater.
- At least one tissue culture incubator dedicated to infected cell cultures. Another separate incubator is desirable for growing uninfected cells.
- The minimal equipment to handle adenovirus culture without exiting the BL2 lab (such as centrifuges, microscope…).
- A sink for hand washing
- A chemical disinfectant kit or at least a gallon of bleach available for spills
For more information about guidelines, visit this NIH web page.
-
Which precautions should I take while working with adenovirus?
Work with adenovirus must be performed in a BL2 lab. There you must:
- Always wear a lab coat while in the virus lab. Before exiting the laboratory for non-laboratory areas (e.g. cafeteria, library, administrative offices…), remove your lab coat and leave it in the laboratory.
- Avoid skin contamination with the virus. Always wear gloves (one pair OK, two pairs better for added protection). Once your gloves have been in contact with infectious material, do not touch common appliances such as telephone or doors handles. Change your gloves frequently.
- Keep the lab doors closed while work is in progress.
- Use mechanical pipetting devices. Do not pipet by mouth.
- Decontaminate all work surfaces after you finish your work, and immediately after any spill. Spray a 10% bleach solution, wipe and spray again a 70% ethanol solution. For large liquid spills, add directly concentrated bleach to the liquid, leave for at least 5 minutes, and wipe.
- Perform all procedures with infectious particles in the biosafety cabinet to minimize the exposure of personnel to aerosols. Minimize the creation of aerosols by pipetting virus cultures and suspension very gently. Use aerosol-resistant tips for pipetting virus suspensions. Do not conduct work with infectious materials in open vessels on the open bench.
- Use needle-locking syringes or disposable syringe-needle units for the injection or aspiration of infectious fluids. Extreme care should be used to avoid auto-inoculation and aerosol generation. Needles should not be bent, sheared, replaced in their sheath or guard or removed from the syringe following use. The needle and syringe should be decontaminated by pipetting in and out concentrated bleach a few times and then promptly placed in a puncture-resistant container.
- Decontaminate all contaminated liquid or solid wastes before disposal. Before starting your virus work, pour some bleach into a beaker. Rinse all materials (tissue culture dishes, pipets, tips…) that came into contact with adenovirus with 10% bleach inside the hood before discarding them in the Biohazard trash and autoclaving. Place all materials to be decontaminated off-site in a durable leakproof container which is closed before removal. If possible, leave the contaminated materials in contact with bleach for a few hours before autoclaving (e.g. after rinsing your pipets with concentrated bleach inside the hood, soak them in a cylinder containing 10% bleach before autoclaving).
- Do not leave the BL2 laboratory with live viruses, unless they are in a sealed tube. Cell cultures transduced with adenoviruses should be inactivated either chemically or biochemically before leaving the BL2 facility.
- Store your adenovirus preparations at –70 °C in closed containers labeled with Biohazard warning signs.
- Wash your hands when exiting the laboratory.
| Vector Map | Polylinker | Sequence | Sequence + Annotations | Product Information Sheet |
|---|---|---|---|---|
| Vector Map | pE3-1_MCS.png (380.64 KB) | pE3-1.txt (2.31 KB) | pE3-1.gb (4.56 KB) | Product_Information_pE3-1.pdf (218.85 KB) |